Recombinant English plantain allergen Pla a 1 Protein
| Cat.No. : | Pla a 1-35E |
| Product Overview : | This recombinant protein contains full length of Pla a 1, a major allergen of English plantain without tag was expressed in E. coli. |
- Specification
- Gene Information
- Related Products
- Case Study
- Application
- Download
| Species : | English plantain |
| Source : | E.coli |
| Description : | Pla a 1 a major allergen of English plantain. |
| Form : | Affinity purified, liquid |
| Purity : | >95% pure by SDS-PAGE |
| Applications : | Suitable for use in ELISA, WB and other immune tests |
| Storage : | Upon receipt, quick spin before open vial, aliquot and store at ≤ -20 centigrade. Avoid multiple freeze/thaw cycles |
| Storage Buffer : | PBS, pH 7.3 |
| Preservative : | 0.05% NaN3 |
| Full Length : | Full L. |
| Synonyms | Pla a 1; Putative invertase inhibitor; Pollen allergen Pla a 1; Pla a 1.0101 |
| UniProt ID | Q8GT41 |
| ◆ Recombinant Proteins | ||
| Pla a 1-35E | Recombinant English plantain allergen Pla a 1 Protein | +Inquiry |
Case 1: Ni WW, et al. Mol Med Rep. 2017
English plantain (Platanus acerifolia) pollen is a prevalent allergen in urban areas, with Pla a 1 being a key component. This research focused on expressing, purifying Pla a 1, and developing monoclonal antibodies against it. The gene was cloned into a pET-28a vector and expressed in ArcticExpress™ (DE3) E. coli. The recombinant protein was used to immunize mice, leading to the creation of hybridoma cells that secreted anti-Pla a 1 antibodies. These antibodies were purified and verified to bind specifically to Pla a 1 in immunoblotting assays, showing potential for diagnostic and therapeutic applications in allergy management.
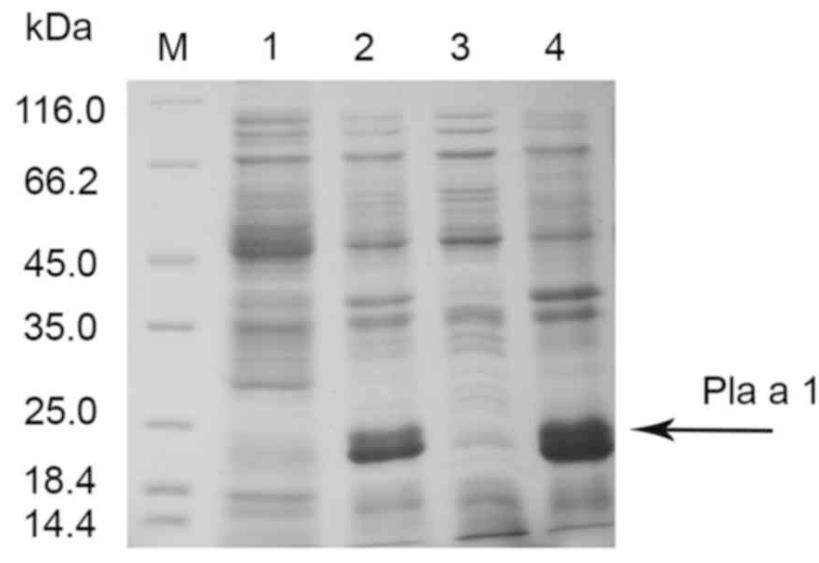
Fig1. SDS-PAGE analysis of the expression of Pla a 1 in Escherichia coli.

Fig2. Immunoreactivity to IgE of Pla a 1 was determined by western blot analysis.
Case 2: Asturias JA, et al. Allergy. 2002
Platanus acerifolia, a prevalent urban tree in the United States and Western Europe, is a major contributor to airborne allergens, yet its pollen's allergenic components remain poorly understood. This study aimed to identify, purify, and characterize the key allergens within this pollen, with a focus on an 18 kDa protein. Using a series of chromatographic techniques, we successfully isolated and characterized Pla a 1, a nonglycosylated protein with a high isoelectric point. Pla a 1 was confirmed as a primary allergen, recognized by 92% of patients with single Platanus allergies and 83% of those with multiple allergies. Further analysis revealed additional allergens at 43 and 52 kDa, with significant IgE-binding prevalences, and profilin, which was particularly notable in patients with multiple sensitivities. This research enhances our understanding of Platanus acerifolia allergens and can inform future diagnostic and therapeutic strategies for allergy sufferers.
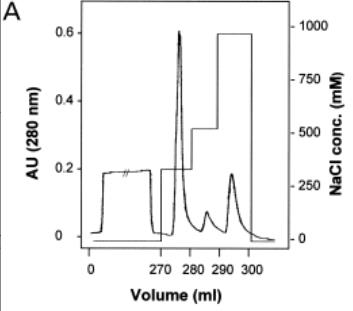
Fig1. Elution profile of P. acerifolia pollen extract on HiTrap SP column.
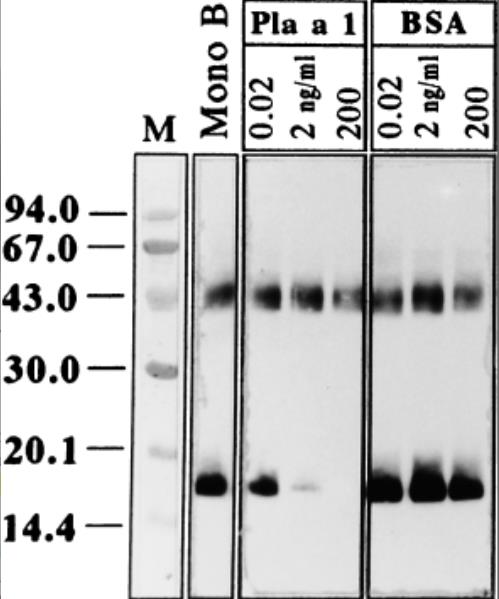
Fig2. SDS-PAGE immunoblotting-inhibition of P. acerifolia pollen extract.
Recombinant English plantain allergen Pla a 1 Protein is a significant component in the field of allergy research, particularly for those allergic to English plantain (Plantanus acerifolia) pollen. This major allergen is a non-glycosylated protein with a molecular weight of approximately 18 kDa and is known to have sequence homology to invertase inhibitory proteins and pectin methylesterase inhibitor proteins. It is one of the key allergens responsible for allergic reactions such as rhinitis, asthma, and atopic dermatitis in many urban areas where the plant is common.
The practical application of recombinant Pla a 1 protein includes its use in the development of diagnostic tools for allergy testing. By expressing and purifying the allergen in a laboratory setting, researchers can create standardized tests to determine the presence and levels of specific IgE antibodies in a patient's serum, which are indicative of an allergic response.
Moreover, the recombinant protein has been instrumental in the production of monoclonal antibodies, which can specifically recognize and bind to Pla a 1. These antibodies are crucial for the development of immunoassays, such as ELISA, which aid in the quantification and detection of the allergen. They also hold potential for the improvement of allergen-specific immunotherapy, a treatment approach that aims to reduce the sensitivity of the immune system to allergens.
In summary, the recombinant English plantain allergen Pla a 1 protein plays a pivotal role in advancing our understanding of pollen allergies and in the development of effective diagnostic and therapeutic strategies for those affected by such allergies.
Not For Human Consumption!
Inquiry
- Reviews
- Q&As
Ask a Question for All Pla a 1 Products
Required fields are marked with *
My Review for All Pla a 1 Products
Required fields are marked with *
Inquiry Basket


